Schedule a Demo
The top-rated Electronic Data Capture system
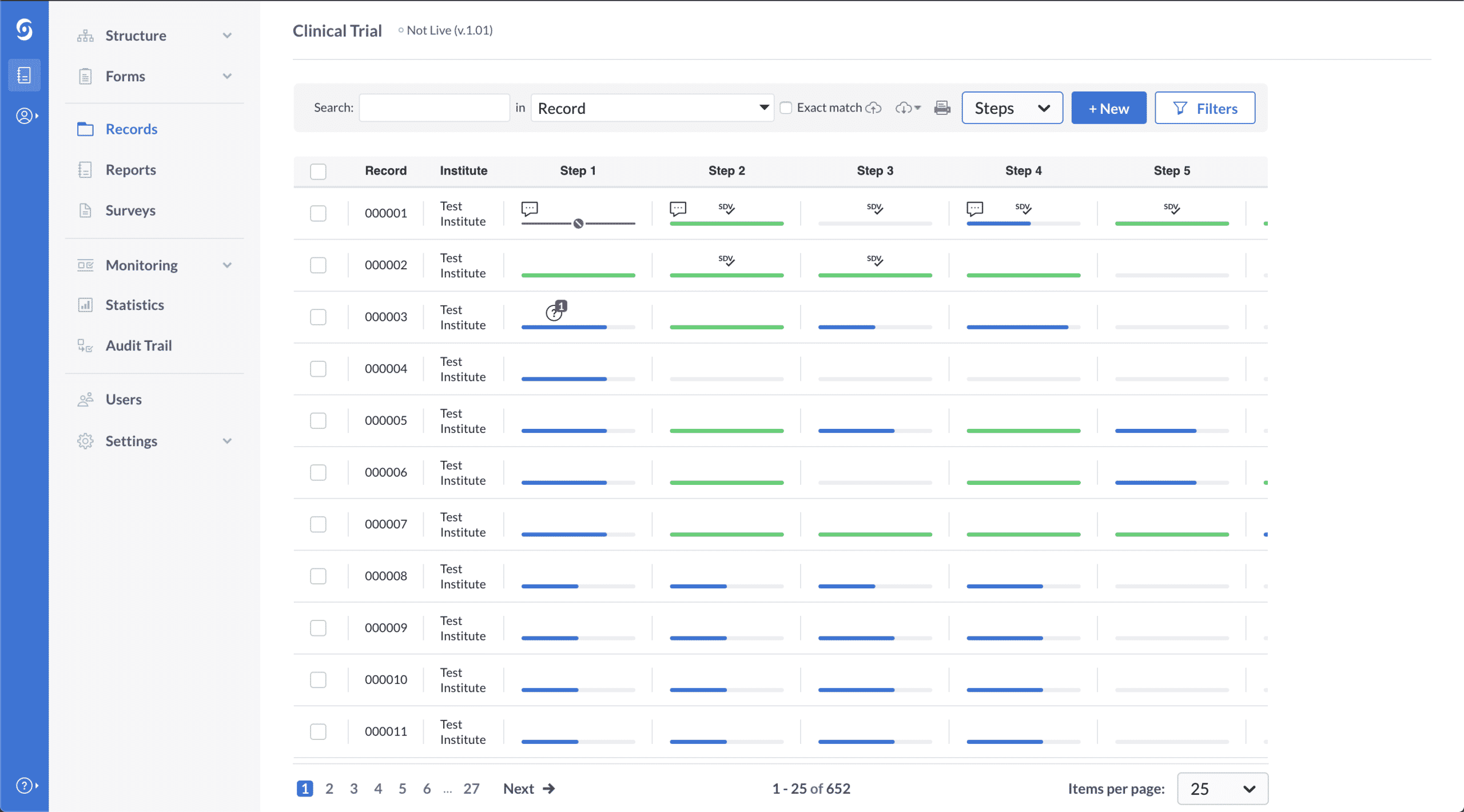
Maximize productivity by easily capturing, processing, and integrating data from multiple sources on a compliant electronic data capture (EDC) system.
Schedule DemoExplore More White Papers

5 steps approach to PMCF to fulfil MDR requirements
Read the white paper to learn how Castor’s 5-step approach to PMCF can be used to fulfill MDR requirements.

What does the MDR tell us about Post-Market Surveillance?
Learn more about the role of Post-Market Surveillance during the lifecycle of your device, the specific requirements for PMS under the MDR, and how an EDC system can help you fulfil these requirements in a cost-effective way.

Three expected changes to the FDA medical device approval process in 2019
At the end of 2018, the FDA announced several upcoming changes to the way medical devices are approved in the United States. In this white paper, we will discuss three expected changes to the FDA medical device approval process in 2019.
Find out how castor can help you with MDR & Post Market Surveillance requirements
Learn more