
Categories
Connect with us on LinkedIn

On-site ePRO in Action: A Recap of Castor’s Product Spotlight
Castor’s on-site ePRO adds in-clinic PRO capture to the ePRO/CDMS workflow you already use. Staff use a site...

EQ-5D in European Trials: When Generic QoL Measures Actually Matter
Analysis of 735 FDA drug approvals reveals 0% EQ-5D labeling inclusion, while European HTA bodies demonstrate 18% technology...
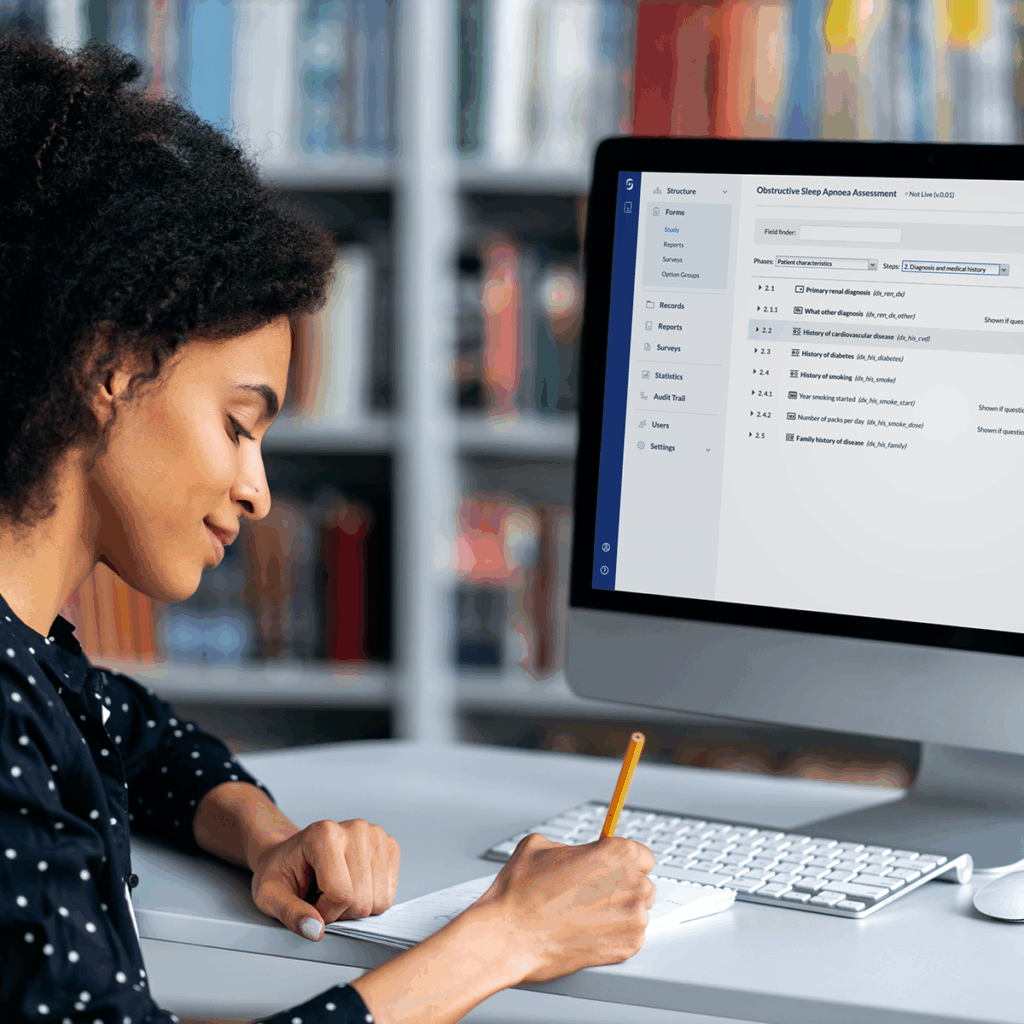
ePRO, eCOA 101: Everything You Need to Know About ePRO and eCOA
Electronic Clinical Outcome Assessment (ECOA) and electronic Patient Reported Outcomes (ePRO) represent far more than digital surveys in...

Why eCOA Still Fails in Clinical Trials: Practical Strategies to Fix Baseline Data Problems
Electronic clinical outcome assessments (eCOA) were supposed to solve data quality and amplify the patient voice. Yet missing...
How a 3-Person Team Got to Phase 3 — Without the Overhead
Gameto proves you don’t need a large CRO or a floor of data managers to run complex global...

The New Standard in Participant-Centric Trials: What Castor’s 2025 Updates Reveal About the Future of Clinical Research
Explore how Castor’s 2025 updates reduce site burden and boost compliance through scalable, flexible DTP workflows.

Do Patients Really Understand Clinical Trials?
Poor comprehension of informed consent disrupts trials. Here’s how to fix it—with data, tech, and actual patient understanding.

Why a Life Sciences Museum Matters—And What It Can Teach Us About the Future of Medicine
What a Museum of Medicine can teach us about trials, trust, and how the public sees (or doesn’t...

Strategies to Minimize Participant and Healthcare Provider Burden in Clinical Trials
This article examines strategies to reduce participant and provider burden in clinical trials while ensuring high-quality data collection....

What do we measure with PROs in head and neck cancer? Pain, QoL, adverse events and more
Patient-Reported Outcomes (PROs) are essential in head and neck cancer research, offering insights into symptoms, quality of life,...

Unlocking FDA’s Vision for PRO Collection in RWE: Timing, Methods, and Insights
Discover FDA's guidance on collecting PROs for Real-World Evidence (RWE), focusing on timing, methods, and improving data quality.

eCOA: Afterthought or Essential Strategy? An Expert’s View on Choosing and Developing an eCOA Strategy
The Evolving Landscape of eCOA in Clinical Trials Clinical trials have long grappled with the complexities of data...

Evaluating “Bother” as a Component of Patient-Reported Outcomes in Clinical Studies
Patient-reported outcome (PRO) instruments refer to multiple scales and questionnaires designed to collect patients’ personal feedback on their...

Revolutionizing Blood Pressure Monitoring: Insights from Aktiia and Castor’s Collaboration
How Aktiia brought their continuous BP device to market by integrating vast amounts of blood pressure data with...

5 Best Practices for Getting Started with ePRO
Electronic Patient Reported Outcomes (ePRO) can become a powerhouse for gathering high-quality data directly from patients. To help...

Can BYOD help us end an era of waste and burden in eCOA?
Bring Your Own Device (BYOD) represents the future of eCOA, and prioritizes flexibility & convenience for trial participants....

Measuring the true patient experience in Cancer Trials with eCOA / ePRO
Shedding light on the complexities and advancements in measuring PROs, drug tolerability, and quality of life in cancer...

The Place of COAs / PROs in the Future of Healthcare
Learn how integrating Patient-Reported Outcomes (PROs) into clinical trials can drive success in Value-Based Care (VBC).